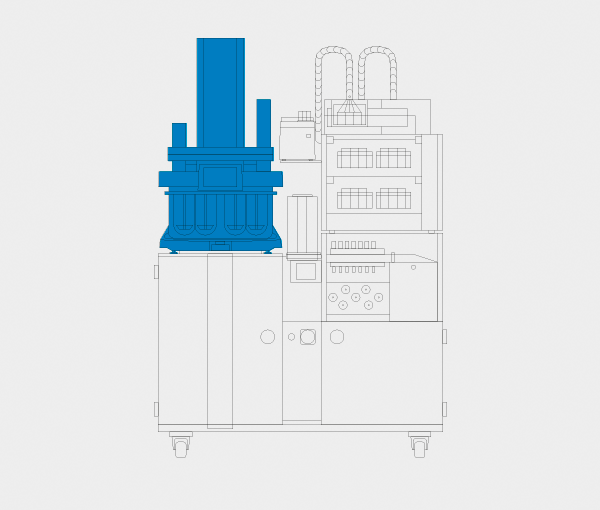
ATF Xtend™ UV On-/Offline
Fully automated dissolution tester with integrated UV-Vis analysis and sample collection.

- 100% FLEXIBLE - Analyze samples immediately with an integrated UV-Vis spectrophotometer or collect them in tubes, vials, and wellplates for offline analysis.
- COMPARE - Automate your Design of Experiment (DOE) studies and easily compare test results of different analytical finishes.
- 100% REPEATABLE - Ensure identical test conditions at all times from media preparation to vessel filling, sampling positions, and filtration.
- ANALYZE - Instant percent dissolved results and real-time dissolution profiles with online UV-Vis measuring using double beam or diode array technology.
- 100% COMPLIANT - Proven SOTAX AutoCompliance™ with USP, Ph.Eur. and other Pharmacopeia requirements.
- DATA INTEGRITY - Automatically execute and record every step of your dissolution test.
- COMPACT - World's most compact fully automated dissolution tester with minimum footprint requirements.
Automation

Highlights

Fully Automated.
Automate your complete dissolution process – from preparing dissolution media and filling vessels to system cleaning at the end of each test run. The ATF Xtend™ automatically prepares the media needed for your dissolution tests, heats & degasses by vacuum (or de-aerates with helium) and precisely fills all vessels including gravimetric verification of delivered volumes. On completion of a test run, all vessels are emptied and the entire system cleans itself with robust customizable washing routines.

Flexibility Maximized.
The ATF Xtend™ combines online UV-Vis measuring and offline sample collection in one compact fully automated dissolution tester. Samples that have been analyzed in the integrated UV-Vis spectrophotometer can be collected for backup or for comparing different analytical finishes. Use of the integrated autosampler is not limited in any way - allowing for safe storage of multi-component products in tubes, capped LC vials, or wellplates from 30-60 timepoints.

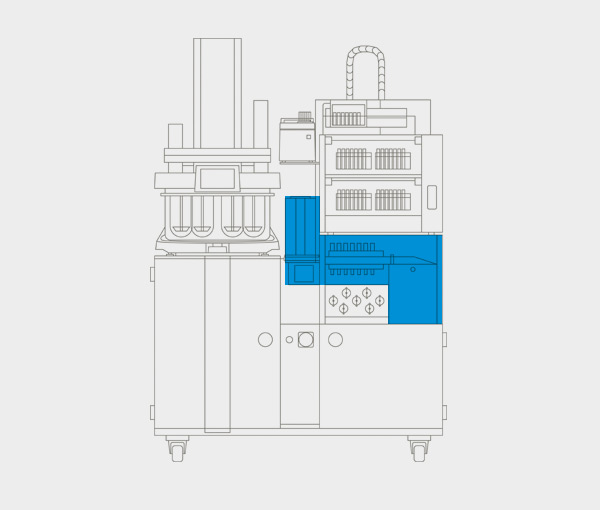
Real-time UV-Vis.
Gather analytical data in real-time and eliminate waiting times by integrating a double beam or diode array UV-Vis spectrophotometer into your sampling process. Withdrawn volumes are immediately returned to the vessels – no medium replacement required. Results including protocolled test conditions are readily available in one consolidated dissolution report at the end of each test run.

Data Integrity.
Methods and recipes are easily managed in full compliance with 21 CFR part 11 requirements, and tests are automatically executed the same way every time. All steps from media preparation to vessel filling, sampling, filtration, and UV-Vis analysis are protocolled. Additionally, CenterView™ video monitoring can be installed to visually record the dissolution process in each vessel. Robust, automated washing routines ensure that there is no carry-over between tests and allow for validation of your cleaning process.

AutoCompliance™
SOTAX dissolution tester are proven to have the lowest wobble rates in the industry. Compliant by design, the ATF Xtend™ with fixed shaft height and self-centering vessels doesn't require any adjustments by the operator. The integrated bath readily accepts the standard-setting MQD™ tool to perform & document mechanical qualification routines according to current FDA guidelines.