History
1991
Integration of Flow-through method in USP.
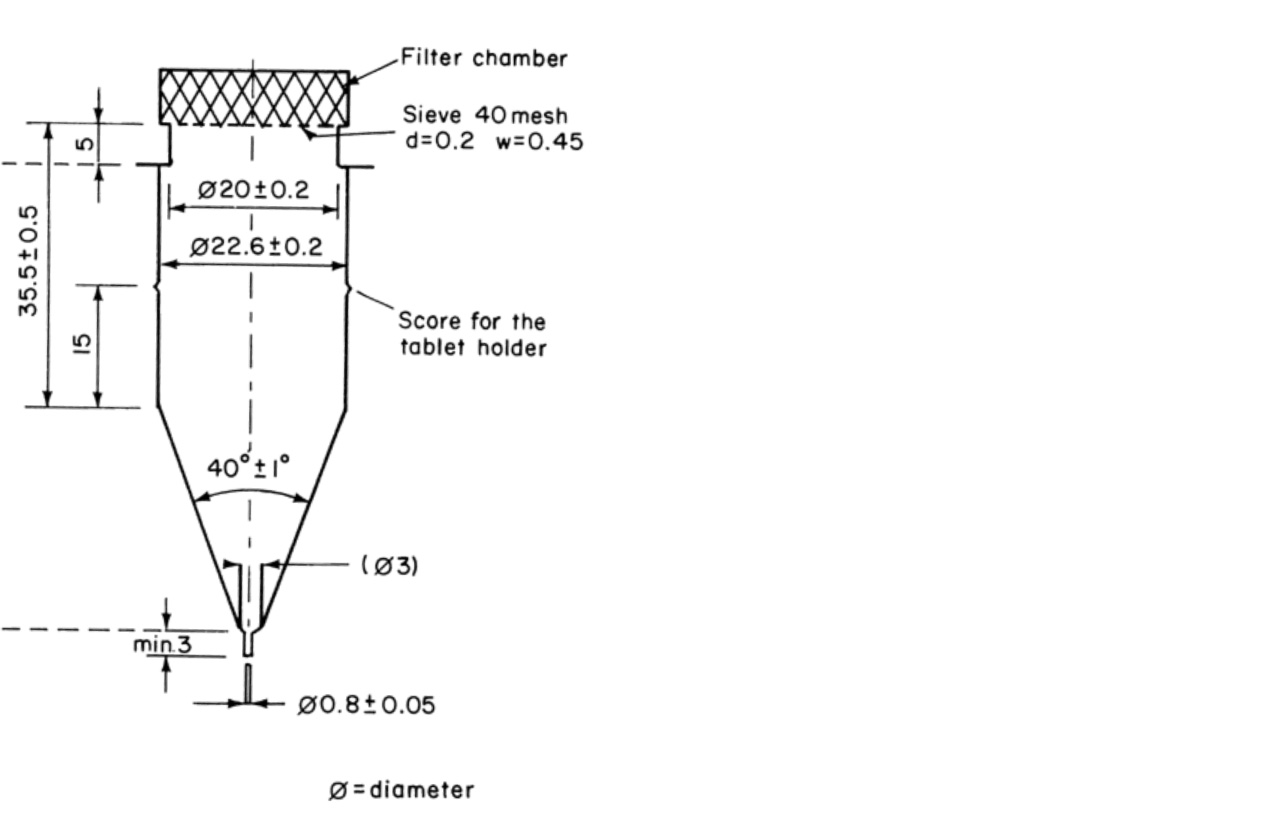
In 1991, the flow-through dissolution method became officially part of the US Pharmacopeia, a widely recognized compendium of pharmaceutical standards. SOTAX played a significant role in the development of this method and its accompanying apparatus. The flow-through cell method is a highly regarded drug release method in the pharmaceutical industry for determining drug quality, and SOTAX is widely recognized as a leader in the field of dissolution and release testing. When people hear the terms "dissolution" or "release testing," they often think of SOTAX.